
Rational Design of New Cryoprotective Agents
Our research program began over 25 years ago with our interest in designing various structural and/or functional "mimics" of antifreeze glycoproteins (AFGPs) that we use as biological probes to study how these compounds control ice growth during freezing/thawing cycle of cryopreservation. Antifreeze Glycoproteins (AFGPs) have the ability to inhibit the growth of ice crystals in vivo. These compounds are essential to the survival of organisms that inhabit subzero environments such as Arctic and Antarctic teleost fish. The structure of a typical AFGP is shown below:
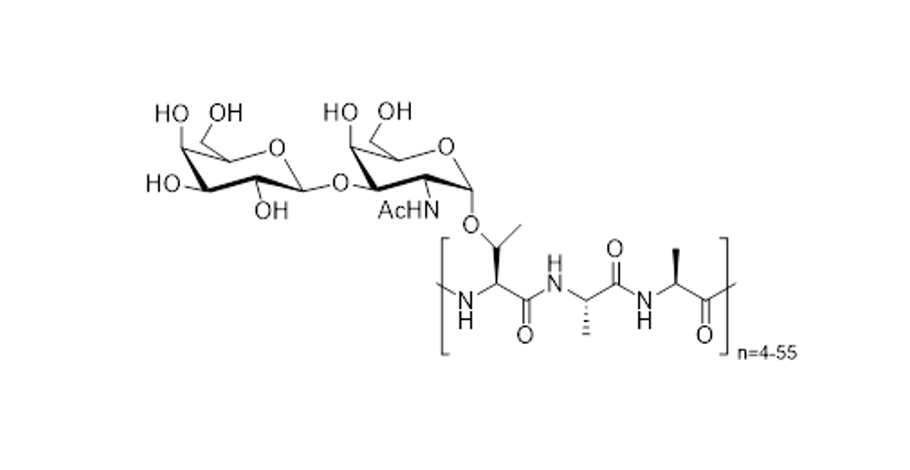
Since the ability to inhibit the growth of ice crystals is very attractive from a practical perspective, these compounds (and structurally related analogues) have potential medical, industrial and commercial applications. Unfortunately, the major source of native AFGP is from polar fish and the isolation and purification of these compounds is a labor-intensive and costly process.
To further elucidate the mechanism by which these glycoproteins function, our laboratory is conducting detailed structure-function studies using various synthetic carbon-linked (C-linked) AFGP mimics. Our synthetic strategy is centered on the preparation of structurally diverse building blocks that are assembled into AFGP mimics using conventional solid phase synthesis. A model illustrating this approach is shown below. This synthesis is general in nature and thus allows for the systematic variation of saccharide structure, amino acid side chain and polypeptide backbone.
The “shift” from glycoconjugates to traditional small molecules as IRIs:
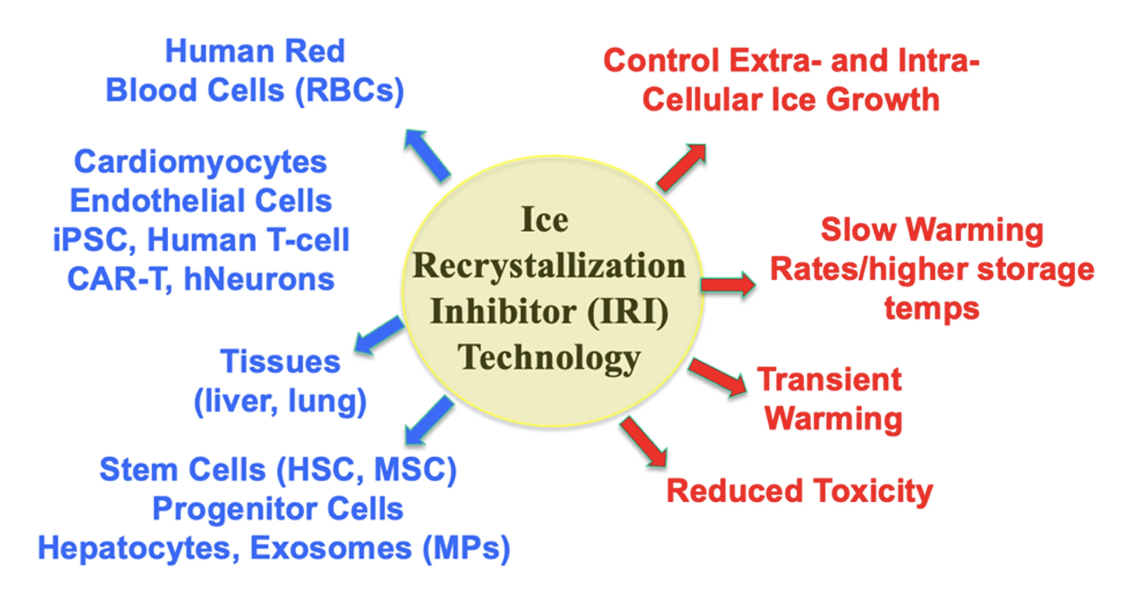
The C-AFGPs designed and synthesized in our laboratory have tremendous potential as cryoprotectants. However, one limitation to these glycoconjugates is their large molecular masses (typically 1.5 to 2.0 kDa) and their lengthy synthesis, which is not amenable to the large-scale production. Following our prior work on C-AFGPs, we have been exploring the possibility of designing small molecules that are potent inhibitors of ice recrystallization. Our initial discovery that hydration of the carbohydrate moiety in C-AFGPs modulated IRI activity was a tremendous scientific contribution to the field and advancement in our laboratory, enabling us to explore a small molecule approach (1,2). Our efforts began with the observation that several mono- and disaccharides were moderate inhibitors of ice recrystallization; however, these were only active in the millimolar range, not the micromolar range associated with the glycoconjugates (2, 3). By correlating the degree of carbohydrate hydration and accounting for the partial molar volume of each carbohydrate, we have devised a method of predicting the IRI activity of a particular carbohydrate. This work has been extended to fluorinated C-linked carbohydrate derivatives (4) and disaccharides (5) have allowed us to make an enormous scientific advancement and move from IRI inhibitors that are complex glycoproteins to truly small molecules. To date, several small molecule amphiphiles have been discovered with IRI activity as in the 0.5 mM range as opposed to the 22-44 mM concentration range (6). The ability to work with small molecules instead of large complex glyconjugates is a tremendous advancement that uniquely positions our laboratory as leaders in this field. The IRIs are an enabling platform technology that offers protection against uncontrolled ice growth, transient warming and a reduction/elimination of conventional cryoprotectants such as DMSO or glycerol.
Learn more about our small molecule CPAs
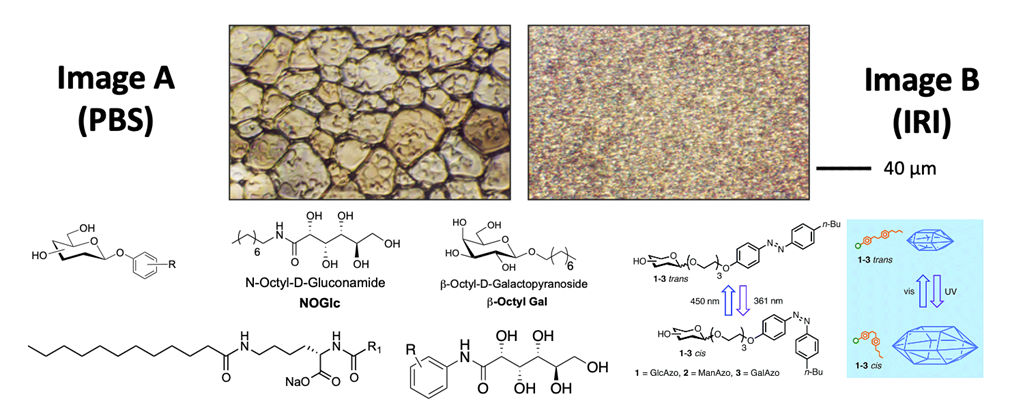
Featured Publications
Czechura, P.; Tam, R. Y.; Dimitrijevic, E. ; Murphy, A. V. ; Ben, R. N. The Importance of Hydration for Inhibiting Ice Recrystallization with C-Linked Antifreeze Glycoproteins. J. Am. Chem. Soc. 2008, 130, 2928. (ACS Highlight article and Faculty of 1000 Biology Editorial highlight)
Tam, R.; Czechura, P.; Ferreria, S. S.; Chaytor, J.; Ben, R. N. Hydration Index – A Better Parameter for Explaining Small Molecule Hydration in Inhibiting Ice Recrystallization. J. Am. Chem. Soc. 2008, 130, 17494. (CIHR, NSERC) (ACS Press Release October 2008)
Chaytor, J. L.; Tokarew, J.; von Moos, E.; Goulla, L.; Findlay, S.; Wu, L.; Allan, D.; Ben, R. N. Inhibiting Ice Recrystallization and Optimization of Cell Viability after Cryopreservation. Glycobiology 2011, 22, 123. (NSERC, Canadian Blood Services)
Chaytor, J. L.; Ben, R. N. Assessing the Ability of a Short Fluorinated Antifreeze Glycopeptide and a Fluorinated Carbohydrate Derivative to Inhibit Ice Recystallization. Bioorg. Med. Chem. Lett. 2010, 20, 5251-5254. (NSERC)
Balcerzak, A.; Ferreira, S. S.; Trant, J. F.; Ben, R. N. Structurally Diverse Disaccharide Analogues of Antifreeze Glycoproteins and their Ability to Inhibit Ice Recrystallization. Bioorg. Med. Chem. Lett. 2012 22, 1719. (NSREC)
Capicciotti, C.; Leclere, M.; Perras, F.; Bryce, D. L.; Paulin, H.; Harden, J.; Ben, R. N. Potent Inhibition of Ice Recrystallization by Low Molecular Weight Carbohydrate-based Surfactants and Hydrogelators. Chem. Sci. 2012 3, 1408. (NSERC, CBS)