
Custom-tailored Antifreeze Activity
Aside from the design of AFGP analogues with enhanced chemical and biological stability, we also seek to design antifreeze glycoconjugates with custom-tailored antifreeze protein-specific activity. Biological antifreezes possess primarily two types of activity. The first is thermal hysteresis which is a depression of freezing point relative to melting point. In conjunction with this, "dynamic ice shaping" is also observed.
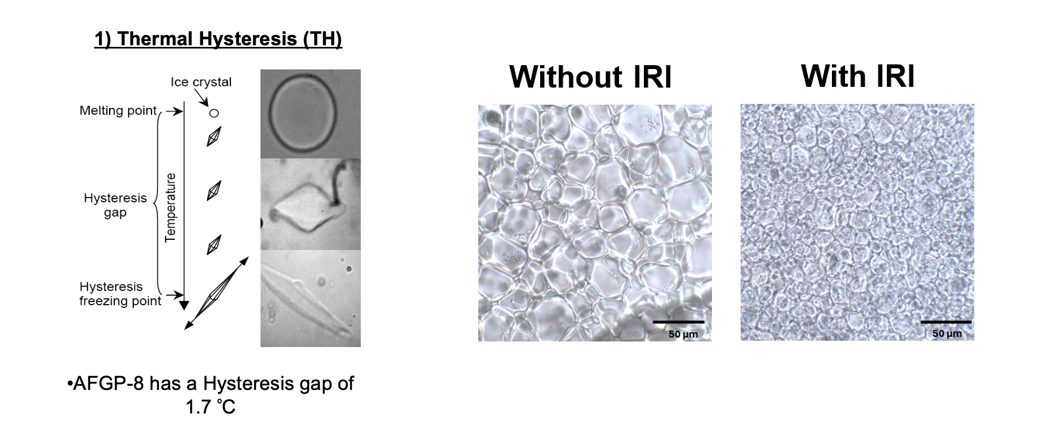
In addition to thermal hysteresis, biological antifreezes also can inhibit recrystallization in frozen samples. While recrystallization-inhibition activity is a very desirable property that makes these compounds attractive as cryoprotectants for cells, tissues and organs, the dynamic ice shaping associated with thermal hysteresis causes significant cellular damage at temperatures outside of the TH gap. Consequently, native antifreeze glycoprotein is a poor cryoprotectant. During the last several years, our laboratory has designed several C-linked AFGP analogues that are extremely potent recrystallization inhibitors but do not posses thermal hysteresis. As a result, these compounds are ideally suited for many cryomedical applications. Our laboratory is presently exploring such applications in various biological systems.
Native antifreeze glycoproteins (AFGPs) have the unusual ability to inhibit the growth of ice in organisms inhabiting sub-zero environments. AFGPs can depress the freezing point of a solution below the melting point in a non-colligative manner. This results in a thermal hysteretic (TH) gap typically between -1 to -8 oC because of the irreversible binding of AFGP to the surface of the ice crystal. This binding event changes the habit or morphology of the ice crystal. AFGPs also have the ability to inhibit ice recrystallization, a process whereby large ice crystals in a frozen sample grow larger at the expense of smaller ice crystals. These two activities are coupled together and this is problematic for cryopreservation applications as the modified ice crystal morphology exacerbates cell damage at the low temperatures associated with cryopreservation. Thus, all biological antifreezes are poor cryprotectants. The ability to inhibit ice recrystallization is highly desirable for cryopreservation applications and our laboratory has successfully “engineered” carbon-linked (C-linked) AFGP analogues that are potent inhibitors of ice recrystallization but do not exhibit TH activity (1,2). We subsequently demonstrated that the carbohydrate moiety in these analogues dramatically modulates IRI activity and highly hydrated carbohydates are the most potent inhibitors of ice recrystallization (3). This work represented a tremendous breakthrough in understanding how these molecules function and has been featured as an ACS highlight article and a Faculty 1000 Biology editorial highlight. Extensive structure activity relationship (SAR) studies have been performed on our C-AFGPs and several structural features (i.e. side chain length, carbohydrate structure, linkage between carbohydrate moiety and peptide backbone) have been identified as important for IRI activity (1,2,4,5).
With the custom-tailored antifreeze activity, these glyconjugates have tremendous potential as cryoprotectants. All of this work was possible because our laboratory developed a new and improved assay for assessing and quantifying recrystallization-inhibition (RI) activity using domain recognition software (6). This assay is the first of its kind that provides a fast and standardized method for analysis of IRI activity permitting the direct comparison of different samples and IRI profiles. The software for this assay was developed in collaboration with the National Research Council and we receive approximately 5-10 requests each year from research labs around the world to have access. The software is provided to researchers at no cost. Our work on the rational design of IRI active glyconconjugates has brought international recognition and as experts in this area well collaborate with top research laboratories in Japan, Australia, Europe, United States and Canada. Recently several reviews have been published top journals (ChemBioChem, Chemical Science, Biomacromolecules and Polymer Science). These articles have highlighted the importance and utility of novel inhibitors of ice recrystallization. Much of the pioneering work discussed in these articles was performed in our laboratory.
Featured Publications
Tam, R. Y.; Rowley, C. N.; Petrov, I.; Zhang, T.; Afagh, N. A.; Woo, T.; Ben, R. N. Solution Conformation of C-Linked AFGP Analogues and Modulation of Ice Recrystallization Inhibition Activity. J. Am. Chem. Soc. 2009, 131, 15745-15753.
Czechura, P.; Tam, R. Y.; Dimitrijevic, E. ; Murphy, A. V. ; Ben, R. N. The Importance of Hydration for Inhibiting Ice Recrystallization with C-Linked Antifreeze Glycoproteins. J. Am. Chem. Soc. 2008, 130, 2928. (ACS Highlight article and Faculty of 1000 Biology Editorial highlight)
Chaytor, J. L.; Ben, R. N. Assessing the Ability of a Short Fluorinated Antifreeze Glycopeptide and a Fluorinated Carbohydrate Derivative to Inhibit Ice Recystallization. Bioorg. Med. Chem. Lett. 2010, 20, 5251-5254.
Capicciotti, C.; Leclere, M.; Trant, J. F.; Ben, R. N. Application of Solid Phase Microwave-Assisted Click Chemistry to the Synthesis of C-Linked Triazole Analogues of Antifreeze Glycoproteins. Bioconjugate Chem. 2011 22, 605.