
Understanding the Mechanism of Action
Many different classes of molecules have the ability to inhibit ice recrystallization. These include polymeric molecules such as antifreeze (glyco)proteins (AF(G)Ps) or polyvinyl alcohol (PVA), and carbohydrate˗based, small˗molecule, biocompatible ice recrystallization inhibitors (IRIs) such as those recently reported by Ben et al. Unlike for IRI active polymers, the mechanism of action of small-molecule carbohydrate-based IRIs is unclear and is necessary in order to gain a clear understanding of structure˗function relationships and further improve efficiency.
Three different hypotheses could explain the IRI activity of carbohydrate-based, small-molecule IRIs:
1) The IRI binds directly onto the surface of ice crystals, causing dynamic ice shaping (DIS) and thermal hysteresis. This is consistent with what is observed with polymers such as AF(G)Ps or PVA, where binding to the ice surface results in a localized freezing point depression known as the Gibbs-Thomson (Kelvin) effect.
2) The IRI is incorporated into the ice lattice creating defects, thereby allowing water molecules to move more freely and altering the behavior and structure of ice crystal formation.
3) The IRI limits the ability for water molecules to migrate within the liquid water phase between neighbouring ice crystals, disfavoring the transfer of water from crystal to crystal, and ultimately inhibiting ice recrystallization. This mechanism has been previously suggested for C-AF(G)P analogues and small-molecule surfactant-based IRIs that do not display thermal hysteresis activity or cause DIS.
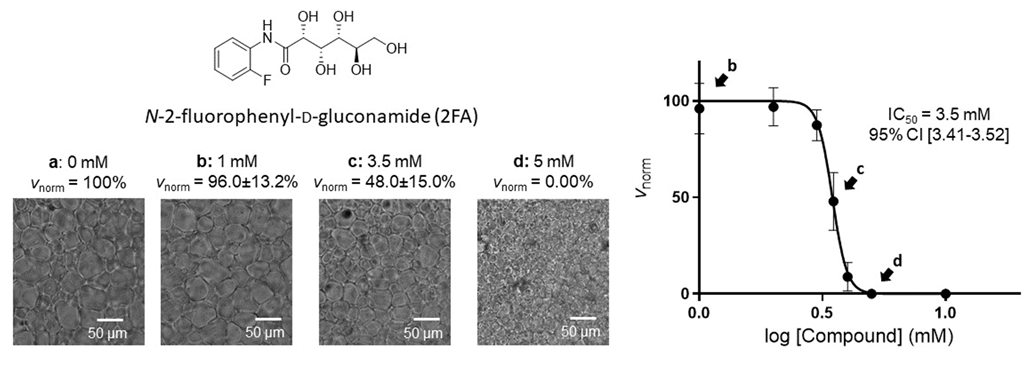
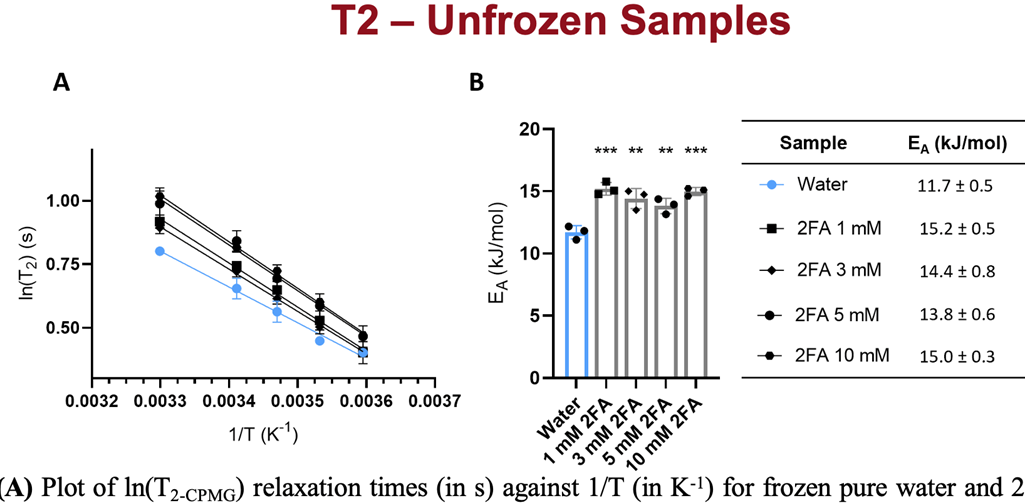
Published work (2023) examined relaxation times of bulk water and frozen water using NMR spectroscopy in the presence of an IRI. Corresponding activation energy of water molecule motion (EAH2O) was calculated, showing that at each concentration 2FA did not affect EA in the solid state, whereas in the liquid state EA was significantly higher with 2FA than for pure water. Therefore, 2FA is excluded from the ice lattice upon freezing and concentrated in the interstitial liquid phase. This restricts the migration of water molecules between ice crystals via the liquid phase, inhibiting ice recrystallization. The results of this study support a mechanism of action in which 2FA increases the energetic barrier for water molecules to move through the liquid phase between ice crystals during the recrystallization process, thus inhibiting ice recrystallization. The results of this study also suggest that the experimental approach may be leveraged to design and screen novel IRI compounds, targeting a maximization of the energetic barrier for water molecule motion within the liquid phase to minimize the effective concentration of IRI for their use in cryopreservation applications. Finally, this study is the first study of its kind, leveraging time˗domain NMR to elucidate the mechanism of action of carbohydrate-based small molecule IRI, and demonstrating its relevance for future studies.
Featured Publications
McMunn, L.E.; D’Costa, A.S.; Bordenave, N.; and Ben, R.N. Probing the Mechanism of Action of Small-Molecule Ice Recrystallization Inhibitors Using Proton Nuclear Magnetic Resonance Relaxation. The Journal of Physical Chemistry Letters 2023 14 (26), 6043-6050